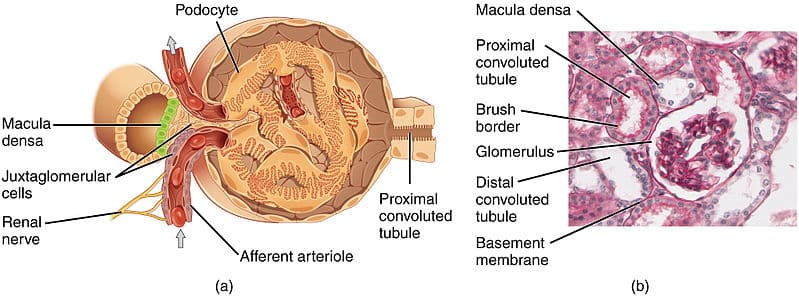
The glomerulus is a loop of capillaries twisted into a ball shape, surrounded by the Bowman’s capsule. This is where ultrafiltration of blood occurs, the first step in urine production. The filtration barrier consists of 3 components:
- Endothelial cells of glomerular capillaries
- Glomerular basement membrane
- Epithelial cells of Bowman’s Capsule (podocytes)
This article shall consider the structure of the filtration barrier, filtration and relevant clinical conditions.
Structure
Endothelial Cells
The glomerular capillary endothelium has many perforations called fenestrae, which are pores about 70nm in diameter. These pores actually do not restrict the movement of water and proteins or large molecules but instead prevent the filtration of blood cells (e.g. RBCs).
Surrounding the luminal surface of the endothelial cells is a glycocalyx consisting of negatively charged glycosaminoglycans. This functions to hinder the diffusion of negatively charged molecules by repelling them due to like charges.
Glomerular Basement Membrane
The basement membrane surrounds the capillary endothelium and is mostly made up of type IV collagen, heparan sulfate proteoglycans and laminin. In particular, heparan sulfate proteoglycans help restrict the movement of negatively charged molecules across the basement membrane.
The basement membrane consists of 3 layers:
- An inner thin layer (lamina rara interna)
- A thick layer (lamina densa)
- An outer dense layer (lamina rara externa)
These layers help to limit the filtration of intermediate and large sized solutes.
Epithelial Cells
Podocytes are specialised epithelial cells of Bowman’s capsule which form the visceral layer of the capsule.
Foot-like processes project from these podocytes and interdigitate to form filtration slits. These filtration slits are bridged by a thin diaphragm (the slit diaphragm) which has very small pores. The pores prevent large molecules, such as proteins, from crossing. Similar to the endothelial cell glycocalyx, negatively charged glycoproteins cover the podocytes, restricting filtration of large anions.
Ultrafiltration
In the glomerulus, blood filters into the Bowman’s capsule in a process called ultrafiltration. Ultrafiltration is simply filtration that occurs under pressure. In this case, the afferent and efferent arterioles are responsible for generating pressure. The afferent arteriole (at the proximal glomerulus) dilates, while the efferent arteriole (at the distal glomerulus) constricts. This creates a pressure gradient throughout the glomerulus, causing filtration under pressure.
The filtration rate of molecules of the same charge across the filtration barrier is inversely related to their molecular weight. Small molecules like glucose (180 Da) are freely filtered whereas albumin (69 kDa) is barely able to cross the barrier. The electrical charges on molecules also play a role in affecting their filtration rate. Negatively charged large molecules filter less easily than positively charged ones of the same size.
Clinical Relevance
Minimal Change Glomerulonephritis
Nephrotic syndrome is a triad of symptoms: proteinuria, hypoalbuminemia and oedema.
Minimal change disease is responsible for 10%-25% of cases of nephrotic syndrome. In minimal change disease, glomeruli appear normal under a light microscope but pathology of the podocytes can be detected under an electron microscope. There is diffuse effacement of the podocytic foot processes, causing widening of filtration slits and visible microvillous changes.
The pathology is still unclear and considered idiopathic. However, it is thought to be due to a T-cell-derived factor. Most patients respond well to steroids but, symptoms may relapse if they come off steroid therapy. Some patients become steroid dependant but most do not progress to chronic renal failure: those that do usually have focal segmental glomerulosclerosis as well.
There are also other forms of glomerulonephritis as well, such as IgA nephropathy and Goodpasture’s Syndrome
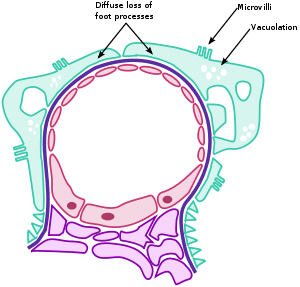
Fig 2 – Diagram showing the main pathologies found within minimal change glomerulonephritis.
Alport Syndrome
Alport Syndrome is an X-linked genetic disease, which causes mutations of the gene coding for α-5 chain of type IV collagen. It presents as progressive chronic kidney disease with haematuria, sensorineural deafness and ocular abnormalities. This results in thinning of the lamina densa in the glomerular basement membrane, with areas of multi-layering producing a basket-weave appearance. In later stages of the disease, glomerulosclerosis, interstitial fibrosis and tubular atrophy occur. There is no curative treatment for Alport syndrome. However ACE inhibitors can reduce progression of renal disease and proteinuria, as well as to control hypertension.