Apoptosis is a physiological process in which cell death is brought about through a regulated sequence of events. It leads to the programmed removal of targeted cells, without harming nearby cells. Defects of this process play an important role in a variety of diseases.
This article shall consider the process of apoptosis, its regulation, and some clinical conditions in which it plays a role.
Apoptosis Pathways
Apoptosis can be initiated by one of two separate pathways; the intrinsic or extrinsic pathway. Both of these end with a final common effector pathway, known as the execution phase.
Intrinsic Pathway
The intrinsic pathway triggers apoptosis in response to internal stimuli:
- Biochemical stress
- DNA damage (this activates the p53 gene – which halts the cell cycle and initiates DNA repair. If this repair attempt is unsuccessful, apoptosis can be induced)
- Lack of growth factors
The intrinsic pathway is modulated by two groups of molecules, Bcl-2 and Bax. Activation of Bax leads to the formation of Bax-Bax dimers, which in turn enhances the action of a variety of apoptotic stimuli – increasing a cell’s susceptibility to apoptosis. The Bcl-2 family consists of both pro- and anti-apoptotic members, and it is the balance between these that determines how susceptible a cell may be to apoptosis.
The balance between the relative concentrations of these groups of molecules establishes a ‘molecular switch’ which determines whether a cell will survive or undergo apoptosis in response to internal stimuli.
Extrinsic Pathway
The extrinsic pathway triggers apoptosis in response to external stimuli, namely by specific ligands binding at ‘death’ receptors on the cell surface.
These receptors are typically members of the Tumour Necrosis Factor Receptor (TNFR) gene family, such as TNFR1 or FAS. Binding at these receptors leads to downstream activation of caspase enzymes.
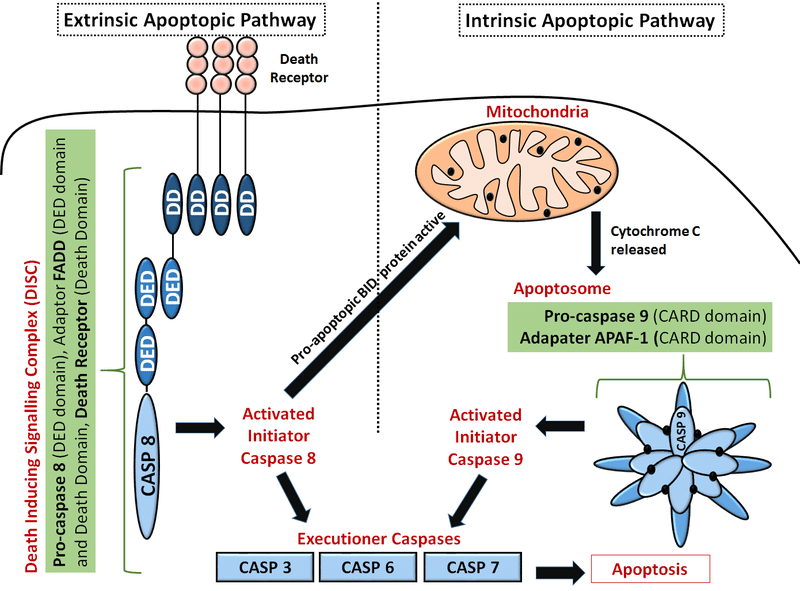
Fig 1 – Diagram showing the intrinsic and extrinsic pathways of apoptosis.
Execution Phase
The initiation of apoptosis by either the intrinsic or extrinsic pathway results in a cascade activation of caspases. These are specialised proteases that normally reside as inactive precursors within the cell.
Initiation of apoptosis first activates initiator caspases, such as caspase 8, the role of which is to cleave other pro-caspases into active ‘executioner’ caspases. These executioner caspases then cause the degradation of a variety of cellular structures, such as the cytoskeleton and nucleus. For example, caspase 3 activates DNAse – leading to fragmentation of DNA.
These processes lead to a variety of morphological changes within the cell, such as nuclear shrinkage (pyknosis) and fragmentation (karyorrhexis). Alongside this, the cell itself shrinks but importantly retains an intact plasma membrane. The dead cells are either immediately phagocytosed by neighbouring cells or break down into smaller, membrane-bound vesicles, known as apoptotic bodies – which are eventually phagocytosed.
Roles of Apoptosis
Aside from providing a regulated form of cell death, for example for cells with irreparable DNA damage, apoptosis plays a major role in embryonic development. Removal of cells via apoptosis is responsible for developing tissue’s form and shape, including:
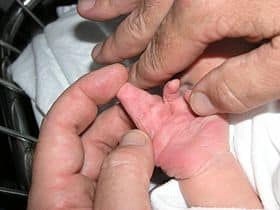
- Interdigital cell death, allowing for separation of the fingers
- Cell death in the urachus, allowing the removal of redundant tissue between the bladder and umbilicus
- Cell death, leading to the removal of vestigial remnants from earlier in development, such as the pronephros
- Removal of the Müllerian or Wolffian ducts when the sex of a fetus is determined
- Cell death in the dorsal part of the neural tube during its closure
- Removal of redundant epithelium following fusion of the palatine processes in the development of the roof of the mouth
If apoptosis fails in these sites, or in another site where it is essential to development, a variety of conditions can develop including syndactyly (webbed fingers), cleft palate, or spina bifida.
Regulation of Apoptosis
There are a variety of factors responsible for regulating apoptosis, which can be intracellular or extracellular. External signals include growth factors or specific signals from other cells, whereas internal factors include DNA damage or failure of cell division.
| Apoptosis Inducers | Apoptosis Inhibitors |
|
|
Clinical Relevance – Syndactyly
Syndactyly is the most common congenital malformation of the limbs and occurs in 1 in 3000 live births. It is a condition in which two or more digits remain fused together after birth, due to a failure of apoptosis.
It can be classified as simple or complex and complete or incomplete depending on the degree of fusion:
- Simple syndactyly – adjacent digits are only joined by soft tissue
- Complex syndactyly – the bones of adjacent digits are also fused
- Complete syndactyly – the skin is fused all the way to the tip of the finger
- Incomplete syndactyly – the skin is only fused part of the way to the fingertip
Syndactyly of the most medial and lateral digits is treated at an early age, to avoid curving of the digits towards each other with growth.